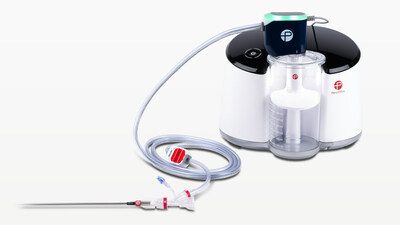
First Patient in STORM-PE RCT Enrolled Evaluating Penumbra’s Latest Computer Assisted Vacuum Thrombectomy – Lightning Flash™ – for Treatment of Acute Pulmonary Embolism
- First-of-its-kind randomized controlled trial comparing computer assisted vacuum thrombectomy (CAVT) using Penumbra’s Lightning Flash™ with anticoagulation versus anticoagulation alone
- Penumbra’s Indigo® Aspiration with Lightning portfolio offers the only CAVT technologies currently available in the U.S. and Lightning Flash is the latest advancement designed to allow physicians to remove large blood clots in the body safely, simply and with speed
ALAMEDA, Calif., Nov. 28, 2023 — Penumbra, Inc. (NYSE: PEN), a global healthcare company focused on innovative therapies, announced the first patient enrolled in STORM-PE, a first-of-its-kind prospective, multi-center, randomized controlled trial evaluating anticoagulation alone vs anticoagulation plus Lightning Flash™ for the treatment of pulmonary embolism (PE). In partnership with PERT Consortium™, a multi-disciplinary group dedicated to improving the care of patients with PE, the STORM-PE trial aims to advance the understanding of the role of computer assisted vacuum thrombectomy (CAVT) in the management of acute PE, with a goal of improving the outcomes for patients with this life-threatening condition.
“STORM-PE is the first head-to-head trial comparing anticoagulation, the mainstay and standard of care for treating acute PE, to CAVT,” said Rachel Rosovsky, MD, MPH, co-global principal investigator of STORM-PE, hematologist and clinical investigator at the Massachusetts General Hospital. “We have already seen the significant impact Lightning Flash can have on patients. The results generated from this pivotal RCT study will provide level 1 evidence of how CAVT compares to current treatment paradigms and will inform us if treatment guidelines need to include this technology as a frontline therapeutic option in patients with intermediate high-risk PE.”
STORM-PE will enroll up to 100 participants at up to 20 sites. Penumbra’s Lightning Flash can be used to remove emboli and thrombi from vessels of the peripheral arterial and venous systems and can be used for the treatment of PE. Lightning Flash combines hypotube-based Indigo Aspiration Catheters with LightningTM Intelligent Aspiration, a unique computer assisted clot detection technology that can differentiate between clot and blood, designed to reduce blood loss and the need for clot-dissolving drugs, which may lower the risk of bleeding complications.
“Enrolling the first patient in STORM-PE RCT is an important milestone,” said Nicolas J. Mouawad, MD, MPH, MBA, RPVI, chief of vascular and endovascular surgery, McLaren Health Care, Bay City, MI. “This landmark RCT will be instrumental to advancing the standard of care for patients with pulmonary embolism.”
“Endovascular therapy has been shown to be an increasingly safe and effective treatment for patients with a symptomatic acute pulmonary embolism,” said Robert Lookstein, MD, MHCDL co-global principal investigator and professor of radiology and surgery at the Icahn School of Medicine at Mount Sinai. “We are proud to commence enrollment in the STORM-PE trial. This trial is designed to answer whether CAVT plus anticoagulation can restore normal function to the failing right ventricle faster and more reliably than anticoagulation alone. We are confident this trial will generate foundational data leading to the next generation of randomized studies in the field of venous thromboembolism for years to come.”
In the U.S., an estimated 900,000 cases of symptomatic PE occur annually[i]. Pulmonary embolism can be life-threatening with 10-30 percent of individuals dying within one month of diagnosisi.
“Our commitment to clinical research and innovation enables us to lead with insight and continue to pioneer interventional therapies that have a significant impact on patients such as our CAVT technologies,” said James F. Benenati, M.D., FSIR, chief medical officer at Penumbra. “In partnership with PERT Consortium, we are committed to generating robust clinical evidence that will help transform care so patients, especially those with serious conditions such as PE, can return home quickly and live fully.”
About Penumbra
Penumbra, Inc., headquartered in Alameda, California, is a global healthcare company focused on innovative therapies. Penumbra designs, develops, manufactures and markets novel products and has a broad portfolio that addresses challenging medical conditions in markets with significant unmet need. Penumbra supports healthcare providers, hospitals and clinics in more than 100 countries. For more information, visit www.penumbrainc.com and connect on Twitter and LinkedIn.
Important Safety Information
Additional information about Penumbra’s products can be located on Penumbra’s website at https://www.penumbrainc.com/providers. Prior to use, please refer to Instructions for Use for complete product indications, contraindications, warnings, precautions, potential adverse events and detailed instructions for use. Risk information can be found at http://www.peninc.info/risk.
Forward-Looking Statements
Except for historical information, certain statements in this press release are forward-looking in nature and are subject to risks, uncertainties and assumptions about us. Our business and operations are subject to a variety of risks and uncertainties and, consequently, actual results may differ materially from those projected by any forward-looking statements. Factors that could cause actual results to differ from those projected include, but are not limited to: failure to sustain or grow profitability or generate positive cash flows; failure to effectively introduce and market new products; delays in product introductions; significant competition; inability to further penetrate our current customer base, expand our user base and increase the frequency of use of our products by our customers; inability to achieve or maintain satisfactory pricing and margins; manufacturing difficulties; permanent write-downs or write-offs of our inventory; product defects or failures; unfavorable outcomes in clinical trials; inability to maintain our culture as we grow; fluctuations in foreign currency exchange rates; potential adverse regulatory actions; and the potential impact of any acquisitions, mergers, dispositions, joint ventures or investments we may make. These risks and uncertainties, as well as others, are discussed in greater detail in our filings with the Securities and Exchange Commission (SEC), including our Annual Report on Form 10-K for the year ended December 31, 2022 filed with the SEC on February 23, 2023. There may be additional risks of which we are not presently aware or that we currently believe are immaterial which could have an adverse impact on our business. Any forward-looking statements are based on our current expectations, estimates and assumptions regarding future events and are applicable only as of the dates of such statements. We make no commitment to revise or update any forward-looking statements in order to reflect events or circumstances that may change.
|
Contact |
|
|
Jennifer Heth |
Parinaz Farzin |
|
Penumbra, Inc. |
Merryman Communications |
|
510-995-9791 |
310.600.6746 |
i “Learn about Pulmonary Embolism,”American Lung Association. Accessed on Oct. 26, 2023. https://www.lung.org/lung-health-diseases/lung-disease-lookup/pulmonary-embolism/learn-about-pulmonary-embolism
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/first-patient-in-storm-pe-rct-enrolled-evaluating-penumbras-latest-computer-assisted-vacuum-thrombectomy—lightning-flash—for-treatment-of-acute-pulmonary-embolism-301998812.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/first-patient-in-storm-pe-rct-enrolled-evaluating-penumbras-latest-computer-assisted-vacuum-thrombectomy—lightning-flash—for-treatment-of-acute-pulmonary-embolism-301998812.html
SOURCE Penumbra, Inc.