-
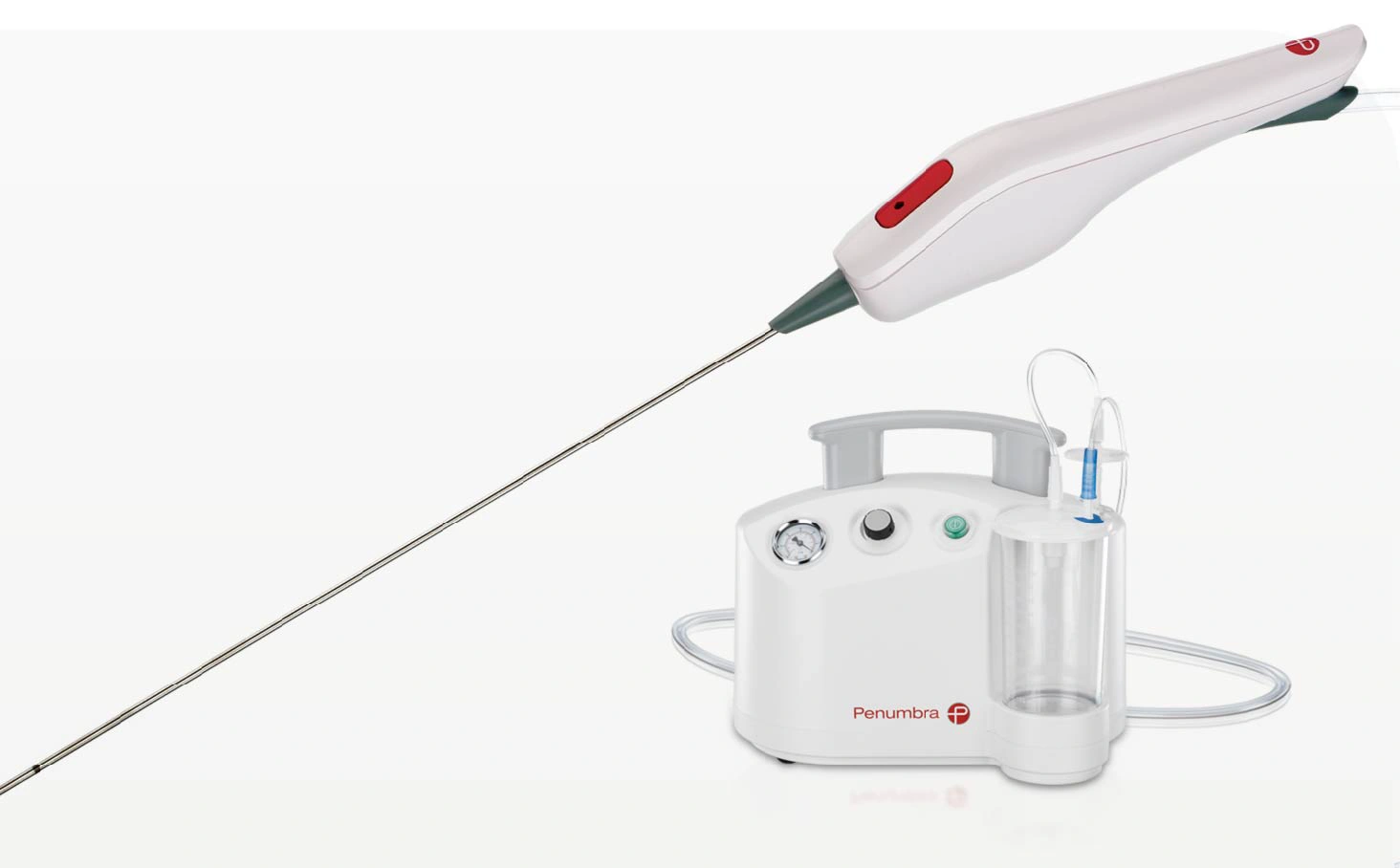
The Artemis™ Neuro Evacuation Device is used for the controlled aspiration of tissue and/or fluid during surgery of the Ventricular System or Cerebrum for patients age 18 or older in conjunction with a Penumbra Aspiration Pump. The Artemis Device works in conjunction with a neuroendoscope through a 19 F (6 mm) sheath. Together with Pump MAX™ Aspiration System, Artemis offers powerful and controlled evacuation.
-
- Minimally invasive cranial access utilising a 14 mm burr hole with 19 F sheath
- Powerful and controlled aspiration up to -29 inHg (or 98.2 kPa)a
- Compatible with neuroendoscopes with working channels 1.6–2.9 mm
- Recessed bident at distal tip maintains cannula patency and aspiration
a. Data on file at Penumbra, Inc.
